
Since phosphate groups can release H + ions into solution, they are considered acidic. Since amino groups can remove H + from solution, they are considered basic.Ĭharged, ionizes to release H +. Since carboxyl groups can release H + ions into a solution, they are considered acidic.Ĭharacterized by central C bound to O and OHĬharged, accepts H + to form NH 3 +. Important Functional Groups in Biologyĭouble bond to oxygen increases the polarityĬharged, ionized to release H +. Other functional groups, such as the carbonyl group, have a partially negatively charged oxygen atom that may form hydrogen bonds with water molecules, again making the molecule more hydrophilic. This carboxyl group ionizes to release hydrogen ions (H +) from the -COOH group resulting in the negatively charged -COO – group this contributes to the hydrophilic nature of whatever molecule it is found on. Among the hydrophilic functional groups is the carboxyl group found in amino acids, some amino acid side chains, and the fatty acid heads that form triglycerides and phospholipids. 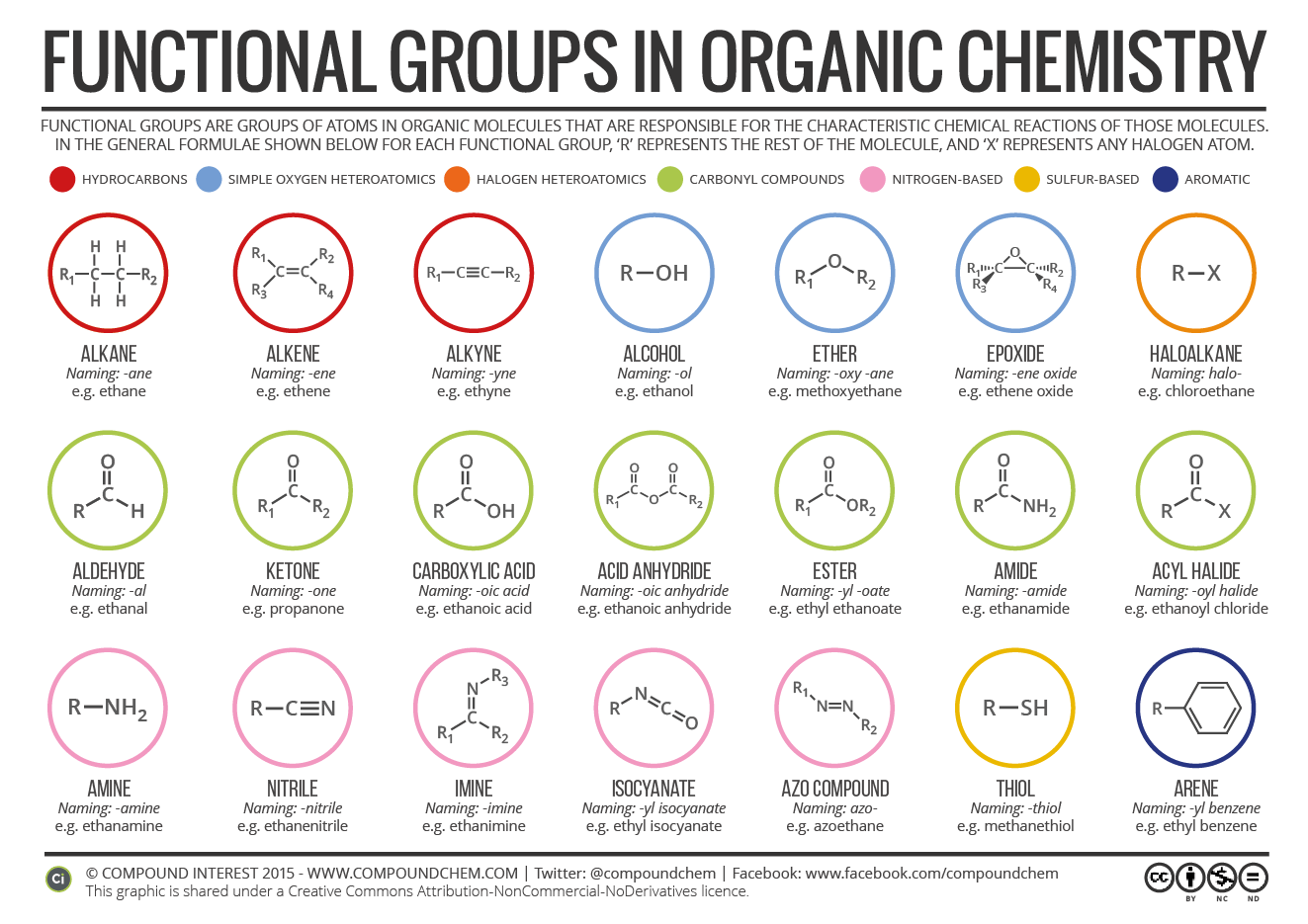
An example of a hydrophobic group is the non-polar methane molecule. lovingly made by Kelly Functional groups are groups of atoms that occur within organic molecules and confer (give) specific chemical properties to those. Classifying Functional Groupsįunctional groups are usually classified as hydrophobic or hydrophilic depending on their charge or polarity. These groups play an important role in the formation of molecules like DNA, proteins, carbohydrates, and lipids. Some of the important functional groups in biological molecules include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl groups. Some of the important functional groups in biological molecules include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl groups. Properties of Functional GroupsĪ functional group can participate in specific chemical reactions. Each of the four types of macromolecules-proteins, lipids, carbohydrates, and nucleic acids-has its own characteristic set of functional groups that contributes greatly to its differing chemical properties and its function in living organisms. Molecules with other elements in their carbon backbone are substituted hydrocarbons. It can stand in for an infinite variety of molecules.įunctional groups are found along the “carbon backbone” of macromolecules which is formed by chains and/or rings of carbon atoms with the occasional substitution of an element such as nitrogen or oxygen. Note: R doesn’t always stand for the same organic molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
